Information
Jul 8, 2024
Multi-Institutional Study of Deep Learning Model for Estimating Pulmonary Function from Chest X-rays
We reported the development and validation of an AI model that estimates forced vital capacity and forced expiratory volume in one second from only chest X-ray images. This model was developed and tested using data from five Japanese institutions, and achieved relatively high accuracy.
Paper
A deep learning-based model to estimate pulmonary function from chest x-rays: multi-institutional model development and validation study in Japan
The Lancet Digital Health
https://doi.org/10.1016/S2589-7500(24)00113-4
Author's Comments
This research addressed a long-standing question of whether pulmonary function could be estimated from chest X-rays, which was actually conceived almost simultaneously with our previous research on deriving cardiac function and valvular disease from images. However, the process of collecting large amounts of data from multiple institutions and establishing a management system took considerable time, requiring more than six years from conception to compiling the results. Additionally, negotiations with reviewers took over a year and there were periods of difficulty in communicating with the editorial board, but we were ultimately able to establish good relationships and this was a significant achievement. After this research was published, we received inquiries from around the world, which has accelerated international collaborative research and partnerships with companies for our laboratory.
Paper Overview
Published in The Lancet Digital Health on July 8, 2024, this study constructed a model that can estimate forced vital capacity (FVC) and forced expiratory volume in one second (FEV1) using deep learning trained on chest X-ray images and spirometry test results collected from five Japanese institutions (A-E) between 2003 and 2021. We analyzed more than 140,000 pairs of chest X-ray and pulmonary function test data, using institutions A-C for training, validation, and internal testing, and institutions D and E for external testing.
Paper Details
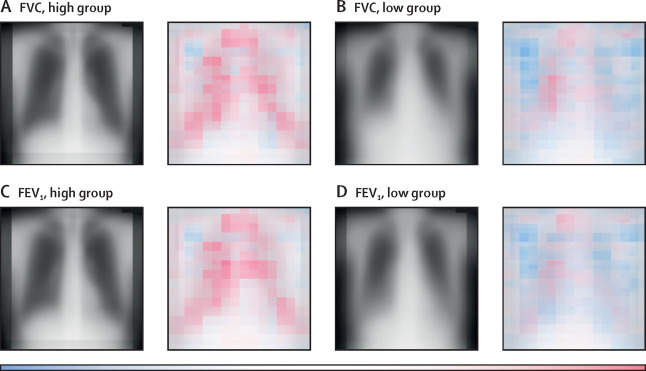
When the developed model was applied to external test data, the correlation coefficient for FVC estimation was 0.91 (99% confidence interval 0.90–0.92) for institution D and 0.90 (0.89–0.91) for institution E. The MSE (mean squared error) was approximately 0.17 L², and the MAE (mean absolute error) was approximately 0.31 L, showing relatively small differences from measured values. For FEV1, the correlation coefficients for institutions D and E were both around 0.91, and the MSE was between 0.11 and 0.13 L², indicating estimation with a certain level of accuracy. Interestingly, the AI model captured multiple aspects of features that reflect respiratory diseases, such as morphological changes in the entire lung and linear shadows in the periphery. This may make it easier to understand the general pulmonary condition even of those who have difficulty undergoing spirometry. However, differences in estimation accuracy due to patient background and types of diseases still exist, and additional validation including a wider range of populations and pathologies is desired. In the future, we would like to continue this research by incorporating clinical information to facilitate the customization of imaging tests and early diagnosis of respiratory diseases.